 How does the pattern of hydrogen bonding explain the lattice that makes up ice crystals? Applications for Hydrogen Bonds Where do hydrogen bonds form? Try changing the temperature of the model. Hydrogen bonds are shown with dotted lines. Interactive: Hydrogen Bonding: Explore hydrogen bonds forming between polar molecules, such as water. Greater electronegativity of the hydrogen bond acceptor will create a stronger hydrogen bond. An electronegative atom such as fluorine, oxygen, or nitrogen is a hydrogen bond acceptor, regardless of whether it is bonded to a hydrogen atom or not. Hydrogen Bond AcceptorĪ hydrogen bond results when this strong partial positive charge attracts a lone pair of electrons on another atom, which becomes the hydrogen bond acceptor. This hydrogen atom is a hydrogen bond donor. In the molecule ethanol, there is one hydrogen atom bonded to an oxygen atom, which is very electronegative. Because of the small size of hydrogen relative to other atoms and molecules, the resulting charge, though only partial, is stronger. The electronegative atom attracts the electron cloud from around the hydrogen nucleus and, by decentralizing the cloud, leaves the hydrogen atom with a positive partial charge. This electronegative atom is usually fluorine, oxygen, or nitrogen. Hydrogen Bond DonorĪ hydrogen atom attached to a relatively electronegative atom is a hydrogen bond donor. This is a space-filling ball diagram of the interactions between separate water molecules. These attractions can occur between molecules ( intermolecularly) or within different parts of a single molecule ( intramolecularly). A hydrogen bond is a type of dipole-dipole interaction it is not a true chemical bond. 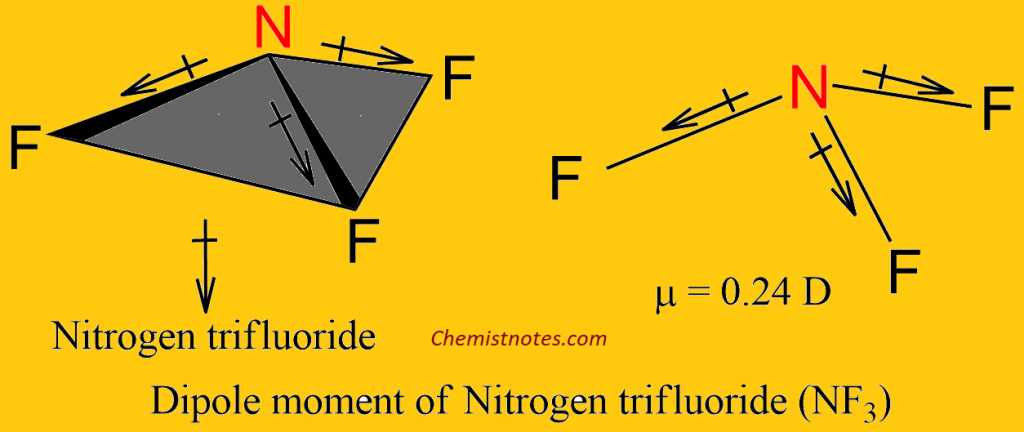
intermolecular: A type of interaction between two different molecules.Ī hydrogen bond is the electromagnetic attraction created between a partially positively charged hydrogen atom attached to a highly electronegative atom and another nearby electronegative atom.hydrogen bond: The attraction between a partially positively charged hydrogen atom attached to a highly electronegative atom (such as nitrogen, oxygen, or fluorine) and another nearby electronegative atom. 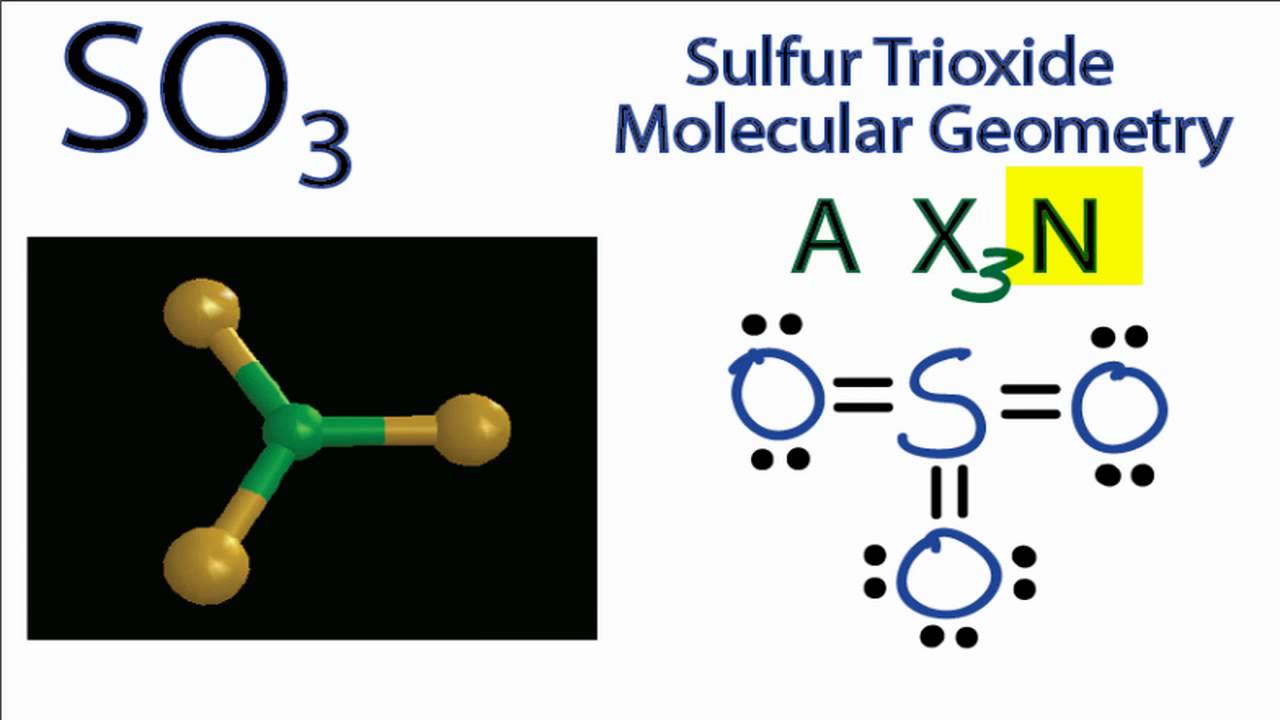
electronegativity: The tendency of an atom or molecule to draw electrons towards itself, form dipoles, and thus form bonds.Hydrogen bonds are responsible for holding together DNA, proteins, and other macromolecules.The hydrogen bond is one of the strongest intermolecular attractions, but is weaker than a covalent or an ionic bond.Greater electronegativity of the hydrogen bond acceptor will lead to an increase in hydrogen bond strength.Hydrogen bonds are strong intermolecular forces created when a hydrogen atom bonded to a highly electronegative atom approaches a nearby electronegative atom.For example, the covalent bond present within a hydrogen chloride (\text_4 has no overall dipole movement. These forces are weak compared to the intramolecular forces, such as the covalent bonds between atoms in a molecule or ionic bonds between atoms in an ionic compound. 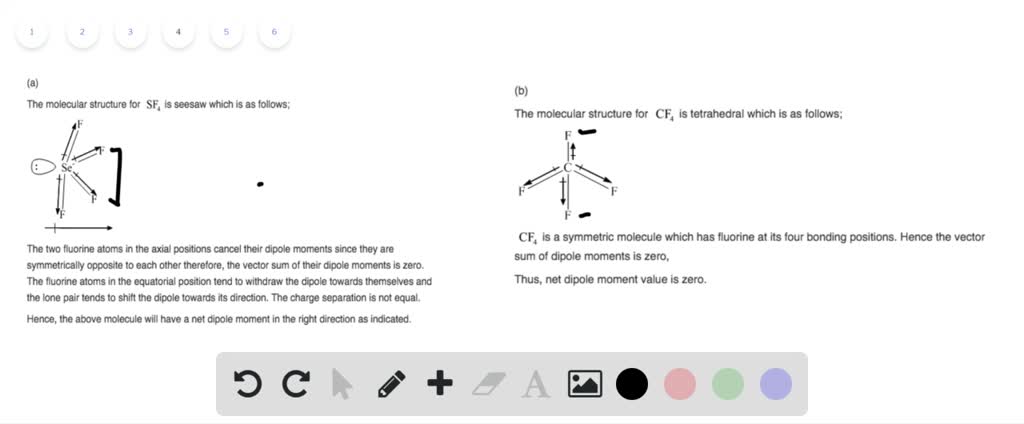
Intermolecular forces are the forces of attraction or repulsion which act between neighboring particles (atoms, molecules, or ions ). Factors that contribute to this include intramolecular dipoles and molecular geometry. polar: In chemistry, a polar molecule is one that has uneven charge distribution.Since each atom has a different affinity for electrons, the ‘push and pull’ of their shared electrons results in one atom maintaining most of the electron density and a partial negative charge, leaving the other atom with a partial positive charge. Dipoles generally occur between two nonmetals that share electrons as part of their bond. dipole: In chemistry, a permanent dipole describes the partial charge separation that can occur within a molecule along the bond that forms between two different atoms. 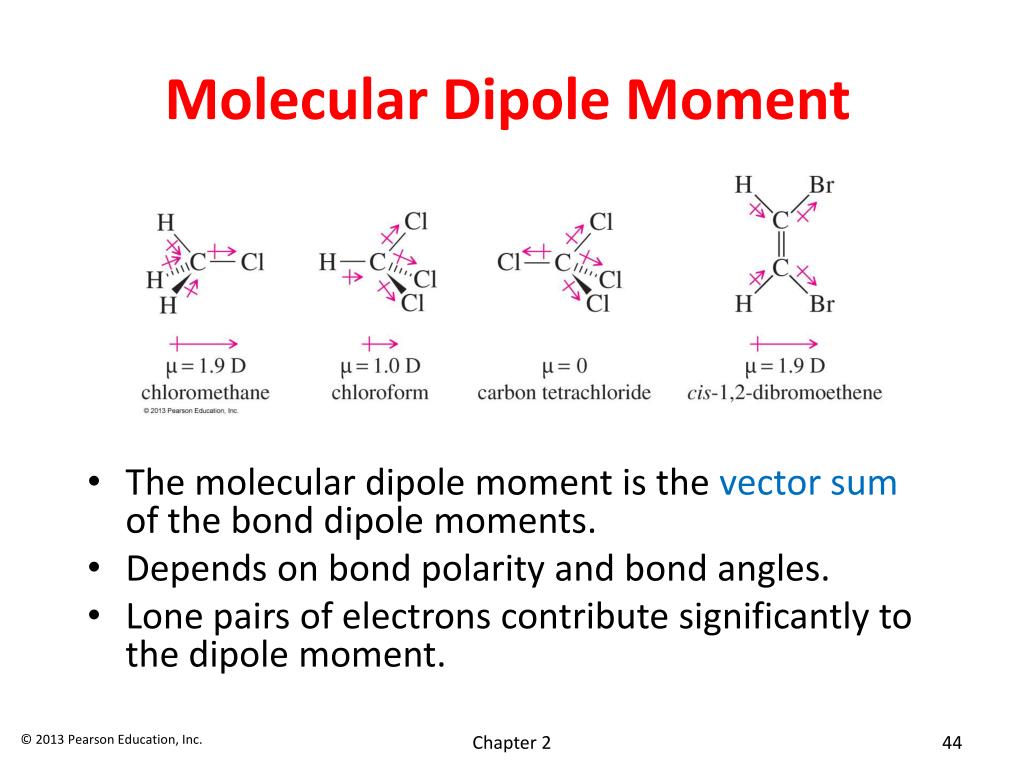
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
